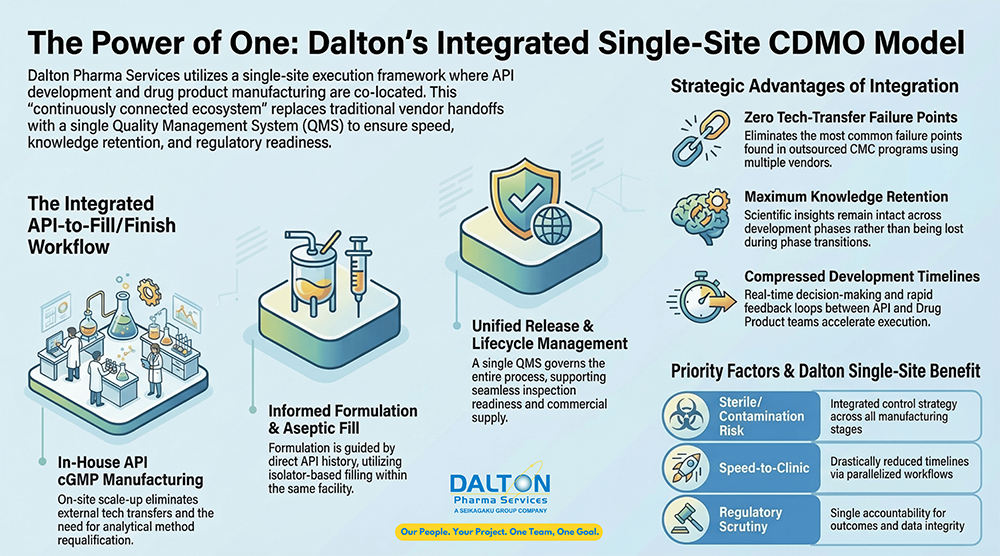
Dalton Pharma Service’s Integrated Single-Site Model
Dalton’s single-site CDMO model is a fully integrated, end-to-end execution framework in which API development, API cGMP manufacturing, formulation development, aseptic drug product fill/finish and full quality control/microbiological testing are co-located within one facility and governed by a single Quality Management System (QMS).\
Rather than operating as a collection of adjacent capabilities, the model is designed as a continuously connected CMC ecosystem, where development decisions, manufacturing execution, and quality oversight are aligned from program inception through commercial supply. This structure preserves process knowledge, eliminates redundant qualification activities, and enables faster, more predictable transitions across development phases.
At its core, the Dalton single-site model replaces sequential vendor handoffs with integrated lifecycle ownership, allowing one organization to remain accountable for technical outcomes, regulatory readiness, and supply continuity.
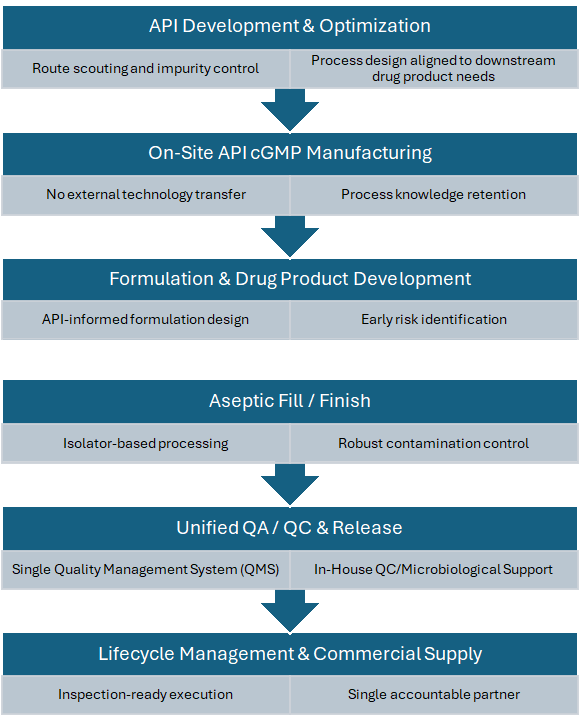
Integrated Process Flow: From API to Sterile Drug Product
Dalton’s process flow is intentionally designed to minimize interfaces, compress timelines, and maintain control over critical quality attributes across the product lifecycle. The integrated process includes API development and process optimization; on-site API cGMP manufacturing;
formulation and drug product development; aseptic fill/finish using modern isolator-based technologies; and unified release and lifecycle management under one quality management system (QMS).
End-to-end process flow:
- API Development and Process Optimization
Early process development, route scouting, and impurity control strategies are executed with downstream drug product and regulatory requirements in mind. Development scientists, analytical teams, and QA operate within the same governance structure, enabling rapid feedback and aligned decision-making.
- API cGMP Manufacturing
Scale-up and cGMP manufacture of the API occur on-site, eliminating the need for external tech transfer packages, re-qualification of analytical methods, or process re-interpretation by a secondary vendor. Process knowledge remains intact as programs transition from development to GMP execution.
- Formulation and Drug Product Development
Formulation development is directly informed by API characteristics and manufacturing history, allowing early identification of risks related to solubility, stability, and compatibility. Integration at this stage reduces late-stage formulation changes and supports smoother validation strategies.
- Aseptic Fill/Finish
Sterile drug product manufacturing is performed within the same facility using modern aseptic technologies, including isolator-based filling and RTU components. Because API and formulation knowledge is retained internally, aseptic processing parameters and contamination control strategies are optimized without re-learning or reinterpretation.
- Release, Lifecycle Management, and Commercial Supply
A unified QC and QA organization oversees batch testing and release, deviation management, change control, and ongoing process verification across API and drug product activities, supporting inspection readiness and lifecycle continuity.
Functional Integration Under One Quality Management System
A defining feature of Dalton’s single-site model is functional integration under a single QMS, rather than parallel systems stitched together through agreements.
Key integration elements include:
- One QMS across API and drug product, aligned with ICH Q7, Q10, and Annex 1 expectations
- Unified change control and deviation management, enabling cross-functional impact assessment
- Integrated contamination control strategy, spanning raw materials, API manufacture, formulation, and aseptic processing
- In-house analytical/microbiological testing and data integrity governance, reducing method drift and documentation gaps
- Embedded QA involvement, supporting real-time decision-making rather than post-execution correction
This structure enables quality to function as a design attribute, not a downstream checkpoint.
Dalton’s Model Reduces Risk and Accelerates Execution
By design, Dalton’s single-site integration removes the most common failure points in outsourced CMC programs.
- No external tech transfers between API and drug product vendors
- No loss of process knowledge during phase transitions
- No duplicated qualification or validation activities
- Faster root-cause analysis and issue resolution, with SMEs on-site
- Clear accountability, with one partner responsible for outcomes
The result is faster execution, greater regulatory confidence, and lower overall program risk, particularly for sterile and complex program
Strategic Fit Within a Broader Outsourcing Strategy
Dalton’s single-site model is particularly well suited for programs where:
- Sterile control and contamination risk are primary concerns
- Speed-to-clinic and timeline are critical
- Regulatory scrutiny is high
- Long-term lifecycle management is anticipated
While selective specialist partnerships may still be appropriate for niche technologies, Dalton’s integrated single-site execution provides a structurally lower-risk default for most API-to-drug product programs.
Dalton Pharma Service’s Integrated Single-Site Model Flowchart |
 |