Reducing Risk and Accelerating API-to-Drug Product Timelines
Integrated Single-Site CDMO Model
Executive Summary
Drug development programs are increasingly constrained by fragmented outsourcing, tight sterile manufacturing capacity, and heightened regulatory expectations. An integrated single-site CDMO model, exemplified by Dalton’s end‑to‑end API and aseptic drug product capabilities, consolidates API development, cGMP manufacturing, and aseptic fill/finish under one Quality Management System (QMS) to reduce tech-transfer friction, preserve critical process knowledge, and compress end-to-end timelines.
This white paper provides an executive decision framework for how a modular single-site facility can deliver speed, flexibility and quality. It also outlines the process controls that de-risk CMC execution from early development through commercial manufacturing.
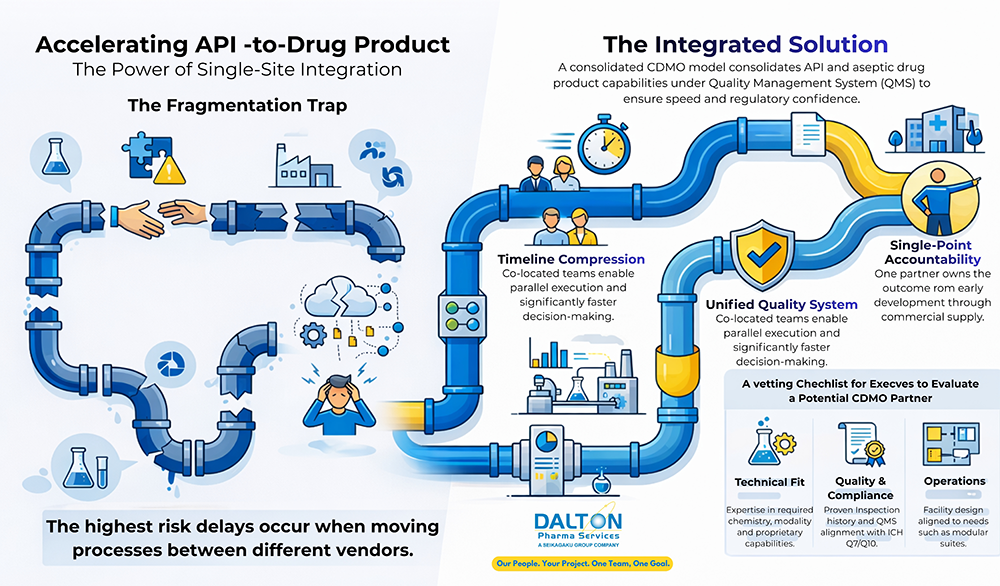
1. The Outsourcing Landscape:
CDMO selection has evolved from a procurement task into an executive decision affecting speed-to-clinic, cost, quality and supply continuity. The market is often framed as a choice between integrated providers and niche specialists. Strategically, integration is most valuable when handoff risk, documentation continuity, and execution predictability are the critical constraints. Dalton’s modular single‑site infrastructure and experienced cross‑functional teams provide partners with predictable execution from early development through commercial supply, aligning speed with regulatory confidence.
2. Why Single-Site Integration Matters
The highest risk delays in CMC programs often occur at the interfaces, moving a process from one vendor to another or from development to manufacturing. Dalton’s single-site model reduces these interfaces by keeping development, API manufacturing, and fill/finish within one facility and one QMS.
- Fewer tech transfers: less data loss and fewer re-qualification cycles.
- Shorter critical path: faster issue resolution with onsite SMEs and QA.
- Higher accountability: one partner owns outcomes from development through drug product.
3. Strategic Advantages of Single-Site Integration
3.1 Executive Positioning
In aseptic fill/finish programs, contamination control, data integrity, and execution predictability are the primary determinants of success. Single-site integration aligns with Annex 1 expectations by consolidating development and GMP manufacturing under one QMS, creating a lower-risk and more inspection-resilient execution model.
3.2 Core Benefits of Single-Site Integration
3.2.1 Elimination of Tech Transfer as a Risk Event
Tech transfers introduce contamination and compliance risk through re-qualification, method bridging, and documentation gaps. Single-site integration removes tech transfer as a recurring lifecycle event by maintaining continuous process and quality ownership.
Net effect:
- Reduced contamination risk during transfers
- Elimination of redundant re-qualification
- Preservation of critical process knowledge
3.2.2 Timeline Compression
Co-located development, QA, and manufacturing teams enable parallel execution and faster decision-making. Timelines are compressed without sacrificing control by aligning development and validation strategies from program inception.
Net effect:
- More predictable speed-to-clinic
- Fewer late-stage corrective actions
- Improved inspection readiness
3.2.3 Unified Quality System
A single QMS simplifies implementation of a holistic Contamination Control Strategy across API and drug product activities. Integrated oversight strengthens change control, deviation trending, and CAPA effectiveness.
3.2.4 Clear Accountability
Single-site integration establishes clear accountability for project and program outcomes, simplifying governance and accelerating high-stakes decision-making.
3.3 Risk Considerations Managed by Design
Common concerns attributed to single-site models, such as flexibility or capacity, are addressed through early feasibility work, modular facility design, and deliberate lifecycle-based redundancy planning.
3.4 Executive Decision Rule
For programs where sterile control, regulatory confidence, and timeline predictability are critical, single-site integration offers a structurally superior execution model.
4. Selection Framework and Vetting Checklist
Selecting the right CDMO requires evidence-based evaluation across technical, operational, quality, business, and relationship dimensions. A practical selection matrix is shown below.
| Pillar | KeyQuestions / Evidence to Collect |
|---|---|
| Technical fit | Demonstrated expertise in the required chemistry, modality, and any differentiated or proprietary capabilities critical to program success. |
| Operations & capacity | Facility design and capacity aligned to program needs, including modular suites, appropriate batch sizes, and modern aseptic processing capabilities (e.g., isolators, RTU, single use). |
| Quality & compliance | Proven inspection history and a sound QMS aligned with ICH Q7/Q10, supported by a robust contamination control strategy and strong data integrity governance. |
| Business stability | Financial strength, clear investment roadmap, and ability to prioritize and sustain clinical programs through key milestones. |
| Partnership | Demonstrated ability to operate as a collaborative partner, with aligned incentives, complementary strengths, and compatible team culture. |
Conclusion
An integrated single-site CDMO model can materially reduce risk and accelerate timelines by minimizing vendor handoffs and consolidating expertise under one QMS. The most resilient outsourcing strategies balance integration benefits with selective specialist partnerships when proprietary technologies or unusually demanding process requirements.
When combined with modular facilities, isolator-based aseptic filling, RTU components, and a sound quality system aligned with ICH and Annex 1 expectations, integrated single-site execution improves predictability from development through drug production while preserving the flexibility to adapt as clinical insights evolve. By embedding contamination control, data integrity, and quality oversight into day-to-day operations, Dalton’s single-site integration model delivers a lower-risk, inspection-resilient approach well suited to sterile manufacturing and complex drug development programs.